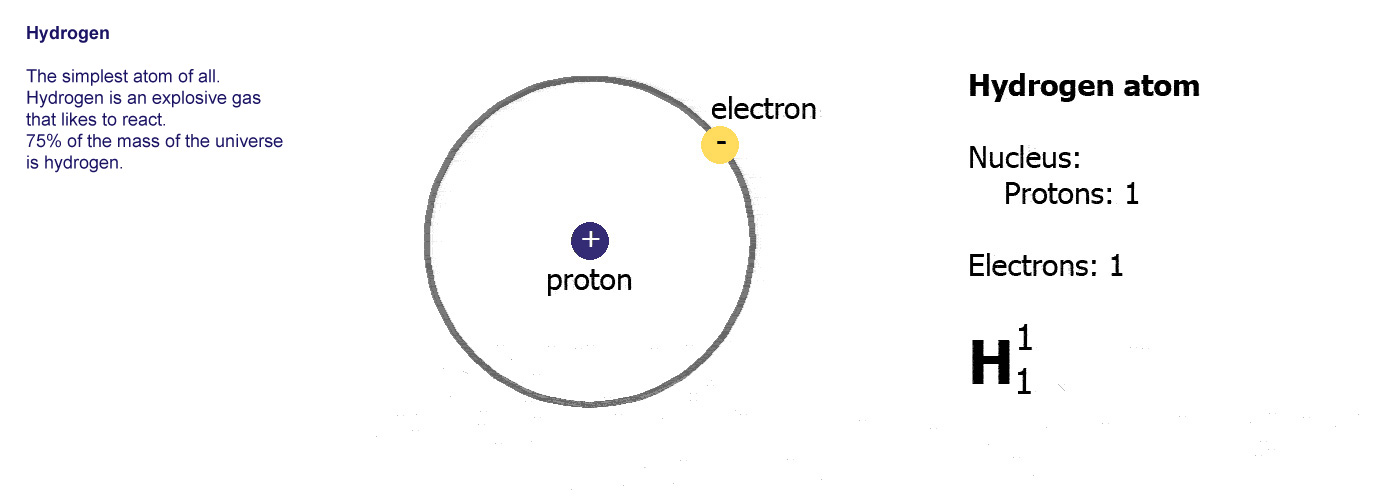
 Because oxygen atoms are electronegative, they attract the shared electrons in covalent bonds. Individual H2O molecules are V-shaped and are made up of two hydrogen atoms that are attached to the sides of a single oxygen atom. How many hydrogen atoms are in a molecule of water? In water, a hydrogen bond is a dynamic attraction between neighboring water molecules that involves one hydrogen atom located between two oxygen atoms. The hydrogen bond is a chemical bond formed by the hydrogen atom and more electronegative elements such as N, O, and F. Check the full article “How many neutrons does hydrogen have?”. Rare hydrogen isotopes, known as deuterium and tritium, have one and two neutrons, respectively. The majority of hydrogen atoms lack a neutron. Hydrogen is the most abundant element in the natureįrequently Asked Questions (FAQs) 1.Deuterium or heavy hydrogen contains one neutron.Natural hydrogen is a simple atom with one proton, one electron, and no neutron.H2 is a colorless, odorless, tasteless, non-toxic, and highly combustible gas.Hydrogen is a diatomic molecule with linear molecular geometry and 180-degree bond angles.īoth hydrogen atoms in the H2 molecule have identical electronegativity, and both atoms share equal ratios of bound shared electrons, resulting in a nonpolar molecule. Hydrogen gas is a loose aggregation of hydrogen molecules, each of which has two atoms, making it a diatomic molecule, H2. The nucleus of a hydrogen atom is made up of one proton with one unit of positive electrical charge and one electron with one unit of negative electrical charge. Hydrogen (H) is the most fundamental member of the chemical element family and is a colorless, odorless, tasteless, flammable gaseous substance. 
Hydrogen is the most abundant chemical substance in the universe, constituting roughly 75 percent of all normal matter. It is colorless, odorless, tasteless, non-toxic, and highly combustible. 
At standard conditions, hydrogen is a gas of diatomic molecules having the formula H 2. Users can continue to use existing He-based mass spectral libraries and quantitative methods.Hydrogen is the chemical element with the symbol H and atomic number 1. Agilent offers a stainless steel install kit (19199S) to help prevent serious contamination problems. The Agilent HydroInert source reduces the need for GC/MS ion source cleaning, decreasing system downtime and maintenance and resulting in fewer interruptions to data generation.Ĭhromatographic-quality stainless steel tubing and fittings are often recommended for hydrogen plumbing. It helps to avoid loss of sensitivity and spectral anomalies, while offering a high-boiler peak shape. The Agilent HydroInert Source is a GC/MS ion source that improves chromatographic performance when using hydrogen as carrier gas in GC/MS. The high price and recurring shortages have increased demand for applications using a hydrogen carrier gas. Helium is a finite resource and this, alongside inefficient production, makes it expensive. Hydrogen carried gas is commonly used as an alternative option to helium. Hydrogen as a carrier gas in GC/MS is a low-cost, renewable gas suitable for several GC/MS applications.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
